Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 18:30
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1

Chemistry, 23.06.2019 00:30
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2

Chemistry, 23.06.2019 01:30
Will a solution form when the solvent and solute are both nonpolar? a. not likely b. never c. most likely
Answers: 1
You know the right answer?
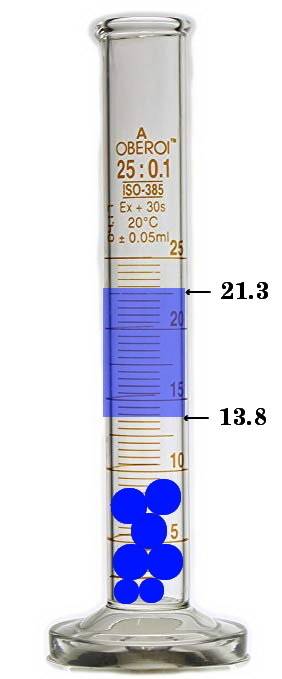
Asample containing 43.98g of metal pellets is poured into a graduated cylinder initially containing...
Questions

Mathematics, 05.05.2020 17:05

Mathematics, 05.05.2020 17:05


English, 05.05.2020 17:05




Health, 05.05.2020 17:05


Biology, 05.05.2020 17:05

Mathematics, 05.05.2020 17:05

Mathematics, 05.05.2020 17:05


Mathematics, 05.05.2020 17:05





Advanced Placement (AP), 05.05.2020 17:05

Mathematics, 05.05.2020 17:05