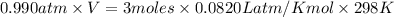Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 14:30
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1

Chemistry, 22.06.2019 21:50
If e is the symbol for an element, which two of the following symbols represent isotopes of the same element? 1. e2. e3. ea.1 and 2c.1 and 4b.3 and 4d.2 and 3
Answers: 2

Chemistry, 23.06.2019 01:00
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1
You know the right answer?
Mg + hcl → mgcl2 + h2 how many liters of hydrogen gas is produced at 298 k and 0.990 atm if 6.00 mol...
Questions







Medicine, 01.12.2021 01:10

Mathematics, 01.12.2021 01:10

Chemistry, 01.12.2021 01:10




Mathematics, 01.12.2021 01:10



Mathematics, 01.12.2021 01:10

Mathematics, 01.12.2021 01:10


Chemistry, 01.12.2021 01:10

Mathematics, 01.12.2021 01:10

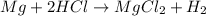
 moles of hydrogen gas that is 3 moles.
moles of hydrogen gas that is 3 moles.