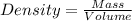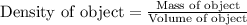Chemistry, 21.06.2019 22:00 NREYESLDS2806
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 23.06.2019 02:50
What is the typical rotational frequency frot for a molecule like n2 at room temperature (25∘c)? assume that d for this molecule is 1å=10−10m. take the total mass of an n2 molecule to be mn2=4.65×10−26kg. you will need to account for rotations around two axes (not just one) to find the correct frequency. express frot numerically in hertz, to three significant figures.
Answers: 3

You know the right answer?
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if...
Questions

History, 16.02.2021 21:20

Mathematics, 16.02.2021 21:20



Mathematics, 16.02.2021 21:20

Mathematics, 16.02.2021 21:20

Physics, 16.02.2021 21:20



Biology, 16.02.2021 21:20


Mathematics, 16.02.2021 21:20

English, 16.02.2021 21:20

Mathematics, 16.02.2021 21:20




Business, 16.02.2021 21:20

History, 16.02.2021 21:20

Mathematics, 16.02.2021 21:20