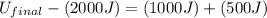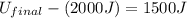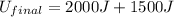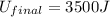In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
In which of these cases are the two wave points considered to be in phase with each other?
Answers: 1

Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 20:30
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2
You know the right answer?
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the f...
Questions





Mathematics, 03.12.2020 07:40

Mathematics, 03.12.2020 07:40

Mathematics, 03.12.2020 07:40

English, 03.12.2020 07:40






English, 03.12.2020 07:40


Advanced Placement (AP), 03.12.2020 07:40



Mathematics, 03.12.2020 07:40

Mathematics, 03.12.2020 07:40

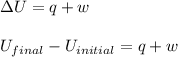
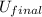 = final internal energy
= final internal energy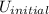 = initial internal energy
= initial internal energy