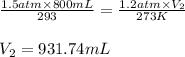Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

You know the right answer?
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation shoul...
Questions

History, 02.12.2019 12:31





Social Studies, 02.12.2019 12:31


Mathematics, 02.12.2019 12:31

Mathematics, 02.12.2019 12:31


English, 02.12.2019 12:31

Mathematics, 02.12.2019 12:31

Mathematics, 02.12.2019 12:31


History, 02.12.2019 12:31



Biology, 02.12.2019 12:31


History, 02.12.2019 12:31

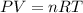
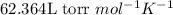
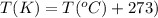
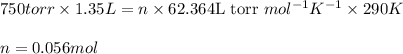
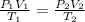
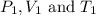 are initial pressure, volume and temperature of the gas
are initial pressure, volume and temperature of the gas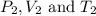 are final pressure, volume and temperature of the gas
are final pressure, volume and temperature of the gas