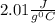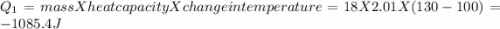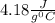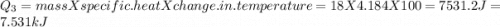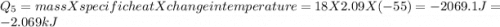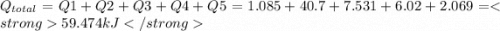Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 14:30
An object resting on a table weighs 100 n. with what force is the object pushing on the table? with what force is the table pushing on the object? explain how you got your answer.
Answers: 3

Chemistry, 22.06.2019 23:00
What is the measured amount of a product obtained from a chemical reaction?
Answers: 1
You know the right answer?
How much heat (in kj) is evolved in converting 1.00 mol of steam at 130.0 ∘c to ice at -55.0 ∘c? th...
Questions

Mathematics, 05.03.2020 20:00













Geography, 05.03.2020 20:03

Mathematics, 05.03.2020 20:04


History, 05.03.2020 20:04



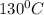 to
to 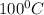 = Q1
= Q1![0^{0}C[/tex=Q3]4) conversion of water to ice=Q45) cooling of ice from [tex]0^{0}C](/tpl/images/0009/5828/c5d88.png) to
to 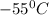 =Q5
=Q5