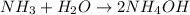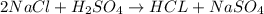Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3
You know the right answer?
Which one of the following chemical equations is balanced? a. 2na + s → 2nas b. koh + h2so4 → khso...
Questions



Biology, 13.04.2021 21:00

Mathematics, 13.04.2021 21:00

Mathematics, 13.04.2021 21:00




English, 13.04.2021 21:00

Mathematics, 13.04.2021 21:00




Mathematics, 13.04.2021 21:00


English, 13.04.2021 21:00

Mathematics, 13.04.2021 21:00


Mathematics, 13.04.2021 21:00

Spanish, 13.04.2021 21:00

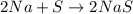
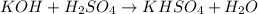
![[(39+16+1)+((2\times 1)+32+(4\times 16))]=154g/mol](/tpl/images/0016/2841/d0994.png)
![[(39+1+32+(4\times 16))+((2\times 1)+16)]=154g/mol](/tpl/images/0016/2841/a82d7.png)