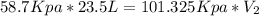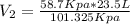Chemistry, 26.06.2019 06:30 carterhoward
23.5 l of h2 is stored at a pressure of 58.7 kpa what volume would the gas take up at stp

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3
You know the right answer?
23.5 l of h2 is stored at a pressure of 58.7 kpa what volume would the gas take up at stp...
Questions

Mathematics, 05.07.2019 00:30

Physics, 05.07.2019 00:30






Mathematics, 05.07.2019 00:30



Biology, 05.07.2019 00:30


Mathematics, 05.07.2019 00:30


Biology, 05.07.2019 00:30


Mathematics, 05.07.2019 00:30


English, 05.07.2019 00:30

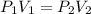
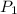 = 58.7 Kpa
= 58.7 Kpa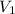 = 23.5 L
= 23.5 L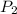 = 101.325 Kpa
= 101.325 Kpa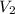 = ?
= ?