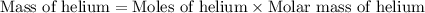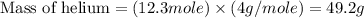Chemistry, 26.06.2019 12:00 dinapearce6078
What is the mass of 12.3 moles of helium (he)?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Drag each number to the correct location on the equation. each number can be used more than once, but not all numbers will be used. balance the equation with the coefficients. 2 3 4 5 kclo3 -> kcl + o2
Answers: 1

Chemistry, 21.06.2019 21:40
It is important to wear proper protective equipment in lab even when not actively performing experiments because accidents can affect any researcher, even one not working on an experiment. select the best answer from the choices provided
Answers: 3


Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1
You know the right answer?
What is the mass of 12.3 moles of helium (he)?...
Questions

History, 13.05.2020 13:57

Mathematics, 13.05.2020 13:57



Biology, 13.05.2020 13:57

Mathematics, 13.05.2020 13:57



Mathematics, 13.05.2020 13:57

Social Studies, 13.05.2020 13:57




Mathematics, 13.05.2020 13:57



Mathematics, 13.05.2020 13:57

Mathematics, 13.05.2020 13:57

Business, 13.05.2020 13:57

Mathematics, 13.05.2020 13:57