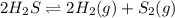Chemistry, 27.06.2019 00:30 cobyontiveros
How does the equilibrium change with the removal of hydrogen (h2) gas from this equation? 2h2s ⇌ 2h2(g) + s2(g) a. equilibrium shifts right to produce more product. b. equilibrium shifts left to produce more reactant. c. equilibrium shifts right to produce less product. d. equilibrium shifts left to produce less reactant.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 1


Chemistry, 22.06.2019 04:30
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

You know the right answer?
How does the equilibrium change with the removal of hydrogen (h2) gas from this equation? 2h2s ⇌ 2h...
Questions

Mathematics, 22.07.2019 17:30

Mathematics, 22.07.2019 17:30






Mathematics, 22.07.2019 17:30



Physics, 22.07.2019 17:30

Mathematics, 22.07.2019 17:30

Computers and Technology, 22.07.2019 17:30




Mathematics, 22.07.2019 17:30

Biology, 22.07.2019 17:30


Physics, 22.07.2019 17:30