
Chemistry, 28.06.2019 09:00 Zachary4759
The enthalpy change for converting 1.00 mol of ice at -25.0 ∘c to water at 90.0∘c is kj. the specific heats of ice, water, and steam are 2.09 j/g−k, 4.18 j/g−k, and 1.84 j/g−k, respectively. for h2o, δ hfus = 6.01kj/mol, and δhvap = 40.67 kj/mol.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1

You know the right answer?
The enthalpy change for converting 1.00 mol of ice at -25.0 ∘c to water at 90.0∘c is kj. the specif...
Questions





Mathematics, 13.04.2020 23:13

Mathematics, 13.04.2020 23:13




Mathematics, 13.04.2020 23:13

History, 13.04.2020 23:13


Mathematics, 13.04.2020 23:13

Mathematics, 13.04.2020 23:13

English, 13.04.2020 23:13





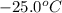 to water at
to water at 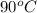 is, 7.712 KJ
is, 7.712 KJ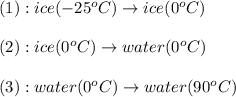
![\Delta H=[m\times c_{ice}\times (T_2-T_1)]+\Delta H_{fusion}+[m\times c_{water}\times (T_3-T_2)]](/tpl/images/0026/6160/eeaad.png)
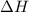 = enthalpy change
= enthalpy change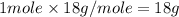
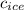 = specific heat of ice = 2.09 J/gk
= specific heat of ice = 2.09 J/gk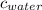 = specific heat of water = 4.18 J/gk
= specific heat of water = 4.18 J/gk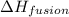 = enthalpy change for fusion = 6.01 KJ/mole = 0.00601 J/mole
= enthalpy change for fusion = 6.01 KJ/mole = 0.00601 J/mole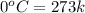
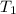 = initial temperature of ice =
= initial temperature of ice = 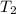 = final temperature of ice =
= final temperature of ice = 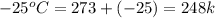
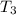 = initial temperature of water =
= initial temperature of water = 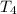 = final temperature of water =
= final temperature of water = 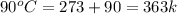
![\Delta H=[18g\times 2.09J/gK\times (273-248)k]+0.00601J+[18g\times 4.18J/gK\times (363-273)k]](/tpl/images/0026/6160/21744.png)
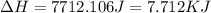 (1 KJ = 1000 J)
(1 KJ = 1000 J)


