
In this reaction, what roll does the lead (ii) nitrate play when 50.0 ml of 0.100m iron (iii) chloride are mixed with 50.0 ml of 0.100m lead (ii) nitrate? a) lead (ii) nitrate increases the amount of precipitate. b) the reactant lead (ii) nitrate decreases product yield. c) lead (ii) nitrate is the excess reactant in the reaction. d) the lead (ii) nitrate is the reaction's limiting reactant.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2


Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2
You know the right answer?
In this reaction, what roll does the lead (ii) nitrate play when 50.0 ml of 0.100m iron (iii) chlori...
Questions





Law, 01.02.2021 21:50



English, 01.02.2021 21:50


Physics, 01.02.2021 21:50

Mathematics, 01.02.2021 21:50


Biology, 01.02.2021 21:50


Mathematics, 01.02.2021 21:50




French, 01.02.2021 21:50

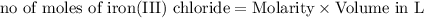
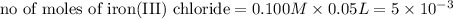
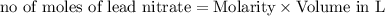
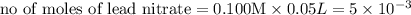
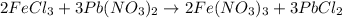
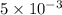 moles of lead nitrate react with
moles of lead nitrate react with 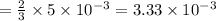 of ferric chloride.
of ferric chloride.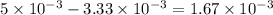 moles of ferric chloride will be left unreacted.
moles of ferric chloride will be left unreacted.

