
When hno3 and au are mixed together, what do you expect to happen, and why? a. no reaction, because au is above h on the activity series and they cannot react b. no reaction, because au is lower on the activity series and cannot replace h c. they form au(no3)2 and h2, because au is more reactive and able to replace h d. they form au(no3)2 and h2, because they trade places in a double replacement reaction

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
As you move from right to left on the periodic table the atomic radius fill in the blank
Answers: 2

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2
You know the right answer?
When hno3 and au are mixed together, what do you expect to happen, and why? a. no reaction, because...
Questions



Mathematics, 21.03.2021 05:10


Mathematics, 21.03.2021 05:10


English, 21.03.2021 05:10


English, 21.03.2021 05:10


Mathematics, 21.03.2021 05:10

Mathematics, 21.03.2021 05:10


Mathematics, 21.03.2021 05:10


Biology, 21.03.2021 05:10


Arts, 21.03.2021 05:10

Spanish, 21.03.2021 05:10

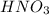 . Thus there will be no reaction.
. Thus there will be no reaction.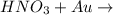 no reaction
no reaction


