
Chemistry, 29.06.2019 05:00 natishtaylor1p8dirz
An atom of sodium-23 (na-23) has a net charge of +1. identify the number of protons, neutrons, and electrons in the atom. then, explain how you determined the number of each type of particle.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
One mole of zinc has a mass of 65.4 grams. approximately how many atoms of zinc are present in one mole of zinc? 32 × 1023 atoms 6 × 1023 atoms 66 atoms 65 atoms
Answers: 1

Chemistry, 22.06.2019 03:00
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1
You know the right answer?
An atom of sodium-23 (na-23) has a net charge of +1. identify the number of protons, neutrons, and e...
Questions


Physics, 20.09.2020 18:01


Chemistry, 20.09.2020 18:01

Arts, 20.09.2020 18:01



English, 20.09.2020 18:01

Medicine, 20.09.2020 18:01

History, 20.09.2020 18:01



Mathematics, 20.09.2020 18:01

History, 20.09.2020 18:01






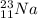
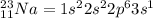
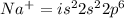 (It will loose one electron)
(It will loose one electron)

