Chemistry, 29.06.2019 22:30 leelee8335
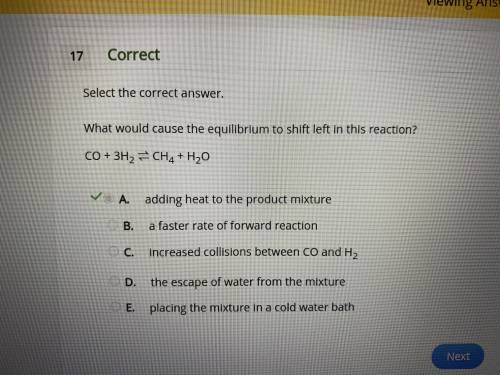
What would cause the equilibrium to shift left in this reaction? co + 3h2 ⇌ ch4 + h2o a. adding heat to the product mixture b. a faster rate of forward reaction c. increased collisions between co and h2 d. the escape of water from the mixture e. placing the mixture in a cold water bath

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3


Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
What would cause the equilibrium to shift left in this reaction? co + 3h2 ⇌ ch4 + h2o a. adding hea...
Questions

Social Studies, 19.07.2019 09:10

Social Studies, 19.07.2019 09:10

Biology, 19.07.2019 09:10

Mathematics, 19.07.2019 09:10

Biology, 19.07.2019 09:10



English, 19.07.2019 09:10


Mathematics, 19.07.2019 09:10

History, 19.07.2019 09:10


Computers and Technology, 19.07.2019 09:10




Social Studies, 19.07.2019 09:10

Social Studies, 19.07.2019 09:10


Mathematics, 19.07.2019 09:10