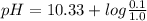Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 23.06.2019 02:00
An alpha particle is: a hydrogen atom a nucleus of helium two neutrons an electron
Answers: 1
You know the right answer?
The pka of hco3 - (or pka2 of h2co3) is 10.33. what is the ph of a solution that has 0.1 m na2co3 an...
Questions


Biology, 19.02.2020 01:50




Mathematics, 19.02.2020 01:51

History, 19.02.2020 01:51




Mathematics, 19.02.2020 01:51






Advanced Placement (AP), 19.02.2020 01:51



Biology, 19.02.2020 01:51

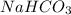 and
and 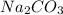 . It acts as a buffer as it is a combination of the weak acid
. It acts as a buffer as it is a combination of the weak acid 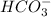 and it's conjugate base
and it's conjugate base 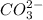 .
.![[Acid] = [HCO_{3}^{-}]=1.0 M](/tpl/images/0034/7560/4bf34.png)
![[Base] =[CO_{3}^{2-}]=0.1 M](/tpl/images/0034/7560/6fbc0.png)
![pH=pK_{a}+log\frac{[CO_{3}^{2-}]}{[HCO_{3}^{-}]}](/tpl/images/0034/7560/044bf.png)