Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 04:30
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
Methanol (ch3oh) has been proposed as an alternative fuel. calculate the standard enthalpy of combus...
Questions

Mathematics, 16.07.2019 10:30

History, 16.07.2019 10:30

Mathematics, 16.07.2019 10:30




Chemistry, 16.07.2019 10:30

Mathematics, 16.07.2019 10:30

Mathematics, 16.07.2019 10:30




Chemistry, 16.07.2019 10:30



English, 16.07.2019 10:30

Chemistry, 16.07.2019 10:30



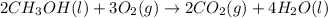
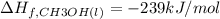

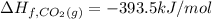
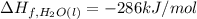
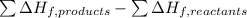
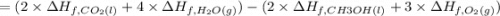
![=[2\times (-393.5 kJ/mol)+4\times (-286 kJ/mol)]-[2\times (-239 kJ/mol)+3\times (0 kJ/mol)]=-1453 kj/mol](/tpl/images/0035/4633/40853.png)