

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

You know the right answer?
Asolution of ammonia and water contains 3.80•10^25 molecules of water and 5.50•10^24 molecules of am...
Questions

World Languages, 17.10.2019 12:30



Social Studies, 17.10.2019 12:30


English, 17.10.2019 12:30

Mathematics, 17.10.2019 12:30

Mathematics, 17.10.2019 12:30

Mathematics, 17.10.2019 12:30

Chemistry, 17.10.2019 12:30

Mathematics, 17.10.2019 12:30




Arts, 17.10.2019 12:30


Chemistry, 17.10.2019 12:30

Mathematics, 17.10.2019 12:30


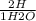 = 7.60×10²⁵atoms of H
= 7.60×10²⁵atoms of H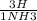 = 1.65×10²⁵atoms of H
= 1.65×10²⁵atoms of H

