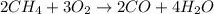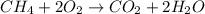Chemistry, 02.07.2019 20:00 annsmith66
Which chemical equation shows the incomplete combustion of methane? a. c3h8 + 5o2 → 3co2 + 4h2o b. 2ch4 + 3o2 → 2co + 4h2o c. ch4 + 2o2 → co2 + 2h2o

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 15:00
Many ionic compounds and a few highly polar covalent compounds are because they completely ionize in water to create a solution filled with charged ions that can conduct an electric current.
Answers: 1

Chemistry, 23.06.2019 00:20
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3
You know the right answer?
Which chemical equation shows the incomplete combustion of methane? a. c3h8 + 5o2 → 3co2 + 4h2o b...
Questions

Mathematics, 14.04.2020 09:52

Chemistry, 14.04.2020 09:52

Biology, 14.04.2020 09:53


Mathematics, 14.04.2020 09:53


English, 14.04.2020 09:53



Mathematics, 14.04.2020 09:54



Mathematics, 14.04.2020 09:54




Chemistry, 14.04.2020 09:55

Biology, 14.04.2020 09:56

Mathematics, 14.04.2020 09:56

Mathematics, 14.04.2020 09:56