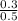Chemistry, 03.07.2019 02:00 710jonathan
Calculate the ph of the solution made by adding 0.50 mol of hobr and 0.30 mol of kobr to 1.00 l of water. the value of ka for hobr is 2.0ă—10â’9. express your answer numerically using two decimal places.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1
You know the right answer?
Calculate the ph of the solution made by adding 0.50 mol of hobr and 0.30 mol of kobr to 1.00 l of w...
Questions


Mathematics, 12.03.2021 19:40

Mathematics, 12.03.2021 19:40


Advanced Placement (AP), 12.03.2021 19:40


Mathematics, 12.03.2021 19:40




Biology, 12.03.2021 19:40





Mathematics, 12.03.2021 19:40

Engineering, 12.03.2021 19:40

Advanced Placement (AP), 12.03.2021 19:40

Biology, 12.03.2021 19:40

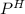 of the solution containing HOBr and KOBr.
of the solution containing HOBr and KOBr.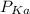 + log
+ log ![\frac{[salt]}{[acid]}](/tpl/images/0044/6662/a0d10.png)
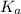 value of HOBr is 2×10⁻⁹.
value of HOBr is 2×10⁻⁹.