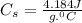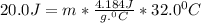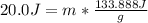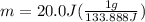Chemistry, 05.07.2019 17:00 BrownieMan123
How many grams of water can be heated from 5.0∘c to 37.0∘c with 20.0j of heat? the specific heat of water is 4.184j/g∘c.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2

Chemistry, 23.06.2019 10:00
An uncovered pot of water lies out in the sun. which statements correctly describe what happens at the surface of the liquid water? 1. the vapor pressure remains constant regardless of the water temperature. 2. the vapor pressure is produced by water molecules that have evaporated. 3. the vapor pressure increases as the sun heats the water in the pot. 4. evaporation stops once the vapor pressure reaches a certain point. 5. evaporation and condensation both occur on the liquid’s surface.
Answers: 3

Chemistry, 23.06.2019 10:10
Solid tin exists in two forms: white and gray. for the transformation sn(s, white) → sn(s, gray) the enthalpy change is -2.1 kj/mol and the entropy change is -7.4 j/(mol*k). a. calculate the gibbs free energy change for the conversion of 1.00 mol white tin to gray tin at -30℃. b. will white tin convert spontaneously to gray tin at -30℃? c. at what temperature are white and gray tin thermodynamically equivalent at a pressure of 1 atm?
Answers: 3
You know the right answer?
How many grams of water can be heated from 5.0∘c to 37.0∘c with 20.0j of heat? the specific heat of...
Questions

Mathematics, 19.07.2019 03:00

Biology, 19.07.2019 03:00



Mathematics, 19.07.2019 03:00


Computers and Technology, 19.07.2019 03:00



Health, 19.07.2019 03:00



Business, 19.07.2019 03:00

English, 19.07.2019 03:00



Mathematics, 19.07.2019 03:00


Health, 19.07.2019 03:00

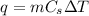
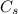 is specific heat and
is specific heat and  is change in temperature.
is change in temperature.