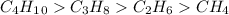Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 21.06.2019 22:50
2. you__turn left on a red light if you are in the left-most lane of a one-way street, you're turning into the left-most lane of a one-way street, and no nearby sign prohibits the turn.
Answers: 2

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2
You know the right answer?
Arrange the molecules in order from strongest intermolecular force to weakest. -c2h6 -c4h10 -ch4 -c3...
Questions


Mathematics, 04.03.2020 01:30



Chemistry, 04.03.2020 01:30

Mathematics, 04.03.2020 01:30

History, 04.03.2020 01:30


Mathematics, 04.03.2020 01:30





Mathematics, 04.03.2020 01:31



Biology, 04.03.2020 01:31

Computers and Technology, 04.03.2020 01:31


Computers and Technology, 04.03.2020 01:31