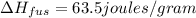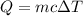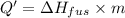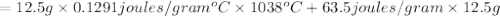Agoldsmith melts 12.4 grams of gold to make a ring. the temperature of the gold rises from 26°c to 1064°c, and then the gold melts completely. if gold’s specific heat is 0.1291 joules/gram degree celsius and its heat of fusion is 63.5 joules/gram, how much energy is gained by the gold? the gold gains a total of joules of energy. i was given this by somebody. no idea what to do with this. qj=(12.4g*(1064-26)°c*0.1291j/g/°c) +(12.4g*63.5j)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:20
One or more substances changing into one or more substances is an example of a
Answers: 1

Chemistry, 23.06.2019 01:30
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1

Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

Chemistry, 23.06.2019 10:00
The temperature of a lead fishing weight rises from 26 °c to 38 °c as it absorbs 11.3 j of heat. what is the mass of the fishing weight in grams?
Answers: 1
You know the right answer?
Agoldsmith melts 12.4 grams of gold to make a ring. the temperature of the gold rises from 26°c to 1...
Questions





Social Studies, 27.10.2020 22:30

English, 27.10.2020 22:30

Mathematics, 27.10.2020 22:30

Social Studies, 27.10.2020 22:30






Mathematics, 27.10.2020 22:30


Biology, 27.10.2020 22:30


Mathematics, 27.10.2020 22:30


Arts, 27.10.2020 22:30