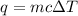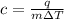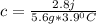Chemistry, 09.07.2019 17:00 jocelynmarquillo1
An unknown metal is suspected to be gold. when 2.8 j of heat are added to 5.6 g of the metal, its temperature rises by 3.9 ∘c.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1

You know the right answer?
An unknown metal is suspected to be gold. when 2.8 j of heat are added to 5.6 g of the metal, its te...
Questions

Mathematics, 20.08.2021 14:00

Social Studies, 20.08.2021 14:00

Physics, 20.08.2021 14:00


Chemistry, 20.08.2021 14:00

Mathematics, 20.08.2021 14:00


World Languages, 20.08.2021 14:00








Mathematics, 20.08.2021 14:00

Business, 20.08.2021 14:00

History, 20.08.2021 14:00


 and it's gold.
and it's gold.