
Chemistry, 10.07.2019 01:30 Gabejenedu
In the combustion analysis of 0.1127 g of glucose (c6h12o6), what mass, in grams, of co2 would be produced?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1
You know the right answer?
In the combustion analysis of 0.1127 g of glucose (c6h12o6), what mass, in grams, of co2 would be pr...
Questions

Chemistry, 21.12.2019 19:31

Computers and Technology, 21.12.2019 19:31



Mathematics, 21.12.2019 19:31





History, 21.12.2019 19:31




Social Studies, 21.12.2019 19:31

Mathematics, 21.12.2019 19:31



Mathematics, 21.12.2019 19:31



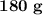 of glucose on combustion gives
of glucose on combustion gives 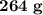 of
of 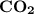 is partial
is partial
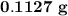 of the glucose on combustion given ......... grams of
of the glucose on combustion given ......... grams of 




