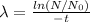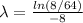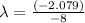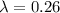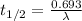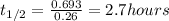Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Identifying limitations of kinetic-molecular theorya chemist is studying the properties of a gas under various conditions. he observes that when the gas is at room temperature and low pressure, it behaves as an ideal gas. when the gas is cooled to 10 kelvin and is placed under high pressure, however, it deviates significantly from an ideal .
Answers: 1

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 01:30
There are main groups in the modern periodic table of elements
Answers: 1

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1
You know the right answer?
Aradioisotope is placed near a radiation detector, which registers 64 counts per second. eight hours...
Questions

Mathematics, 18.11.2020 01:40


English, 18.11.2020 01:40

Mathematics, 18.11.2020 01:40



Advanced Placement (AP), 18.11.2020 01:40

Arts, 18.11.2020 01:40

Physics, 18.11.2020 01:40

Chemistry, 18.11.2020 01:40

Mathematics, 18.11.2020 01:40

Mathematics, 18.11.2020 01:40

Computers and Technology, 18.11.2020 01:40


English, 18.11.2020 01:40

Mathematics, 18.11.2020 01:40

Mathematics, 18.11.2020 01:50

Mathematics, 18.11.2020 01:50

English, 18.11.2020 01:50

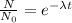
![ln [\frac{N}{N_{0}}]= - \lambda t](/tpl/images/0071/8208/fae60.png)