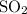Chemistry, 10.07.2019 17:00 kylieweeks052704
Part a choose the pair of substances that are most likely to form a homogeneous solution. choose the pair of substances that are most likely to form a homogeneous solution. co and c6h14 nh2ch3 and ch4 cbr4 and sf2 nf3 and so2 none of the pairs above will form a homogeneous solution.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:30
What type of scientific model does the flow chart represent? (a chart of the scientific process) a. conceptual b. mathematical c. physical d. virtual
Answers: 1

Chemistry, 21.06.2019 22:30
Naoki's bicycle has a mass of 10 kg. if naoki sits on her bicycle and starts pedaling with a force of 168 n, causing an acceleration of 2.8 m/s2, what is naoki's mass?
Answers: 1

Chemistry, 22.06.2019 02:30
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1
You know the right answer?
Part a choose the pair of substances that are most likely to form a homogeneous solution. choose the...
Questions

Biology, 18.10.2019 10:30



History, 18.10.2019 10:30

Social Studies, 18.10.2019 10:30

Advanced Placement (AP), 18.10.2019 10:30




Mathematics, 18.10.2019 10:30

Mathematics, 18.10.2019 10:30

History, 18.10.2019 10:30

History, 18.10.2019 10:30



English, 18.10.2019 10:30



Mathematics, 18.10.2019 10:30

History, 18.10.2019 10:30

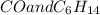 : CO is a polar gas and
: CO is a polar gas and 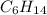 is a non polar liquid. Therefore, this solution will not be homogeneous.
is a non polar liquid. Therefore, this solution will not be homogeneous.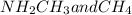 :
: 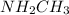 is a polar liquid while methane is a non polar gas. So, this solution will not be homogeneous.
is a polar liquid while methane is a non polar gas. So, this solution will not be homogeneous.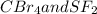 :
: 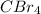 is a non polar solid while
is a non polar solid while 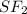 is a polar gas. So, they cannot form homogeneous solution.
is a polar gas. So, they cannot form homogeneous solution.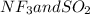 :
: 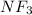 and
and 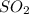 are both gases. So, they form a uniform homogeneous solution. Both can uniformly interact as they both are polar compounds.
are both gases. So, they form a uniform homogeneous solution. Both can uniformly interact as they both are polar compounds.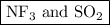 is the pair that is most likely to form a homogeneous solution.
is the pair that is most likely to form a homogeneous solution.
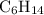
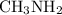 and
and 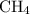
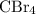 and
and 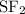
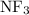 and
and