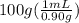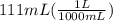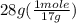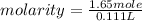Chemistry, 11.07.2019 18:30 aylineorozco836
Commercial concentrated aqueous ammonia is 28% nh3 by mass and has a density of 0.90 g/ml. what is the concentration of ammonia in molarity?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:50
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hydrogen gas and aqueous sodium hydroxide. part a write a balanced chemical equation for this reaction. express your answer as a chemical equation. identify all of the phases in your answer.
Answers: 3

Chemistry, 21.06.2019 22:30
Omg imgonnafailnfiedkla use complete sentences to explain how the mass of hydrogen is conserved during cellular respiration.
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1
You know the right answer?
Commercial concentrated aqueous ammonia is 28% nh3 by mass and has a density of 0.90 g/ml. what is t...
Questions

Social Studies, 29.08.2019 15:20

History, 29.08.2019 15:20


Biology, 29.08.2019 15:20

History, 29.08.2019 15:20

Physics, 29.08.2019 15:20




Mathematics, 29.08.2019 15:20


Mathematics, 29.08.2019 15:20

Business, 29.08.2019 15:20






Mathematics, 29.08.2019 15:20