
Chemistry, 12.07.2019 05:30 myparentsrock17
Achemist adds 220.0ml of a 0.194 m potassium permanganate kmno4 solution to a reaction flask. calculate the mass in grams of potassium permanganate the chemist has added to the flask

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1


Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 22.06.2019 22:30
What must be in balance for temperatures to remain constant?
Answers: 1
You know the right answer?
Achemist adds 220.0ml of a 0.194 m potassium permanganate kmno4 solution to a reaction flask. calcul...
Questions


English, 22.09.2021 14:00


English, 22.09.2021 14:00

Business, 22.09.2021 14:00


English, 22.09.2021 14:00


Mathematics, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00

History, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00


English, 22.09.2021 14:00

Advanced Placement (AP), 22.09.2021 14:00



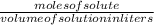 -(1)
-(1)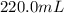 (given)
(given)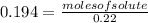
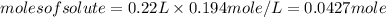
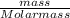 -(2)
-(2)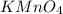 =
= 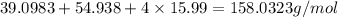
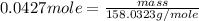
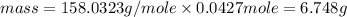
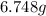 .
.

