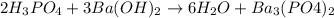Chemistry, 12.07.2019 21:30 hannahmr092402
Identify the neutralization reaction. 2nh3 + h2so4 ⟶ (nh4)2so4 2h3po4 + 3ba(oh)2 ⟶ 6h2o + ba(po4)2 2 no2 ⟶ 2 o2 + n2 po4 3pb + 2h3po4 ⟶ 3h2 + pb3(po4)2

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2

Chemistry, 23.06.2019 00:30
What is the chemical formula of magnesium bromide? a. mgbr2 b. mgbr c. mg2br2 d. mg2br
Answers: 3
You know the right answer?
Identify the neutralization reaction. 2nh3 + h2so4 ⟶ (nh4)2so4 2h3po4 + 3ba(oh)2 ⟶ 6h2o + ba(po4)2 2...
Questions






Geography, 05.10.2019 15:00

Geography, 05.10.2019 15:00










Mathematics, 05.10.2019 15:00


History, 05.10.2019 15:00

Social Studies, 05.10.2019 15:00