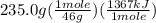Chemistry, 13.07.2019 01:00 marvinc5603
The combustion of one mole of liquid ethanol, ch3ch2oh, produces 1367 kj of heat. calculate how much heat is produced when 235.0 g of ethanol are combusted.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
The combustion of one mole of liquid ethanol, ch3ch2oh, produces 1367 kj of heat. calculate how much...
Questions



English, 03.12.2021 19:50


Mathematics, 03.12.2021 19:50




Social Studies, 03.12.2021 19:50

English, 03.12.2021 19:50








Social Studies, 03.12.2021 19:50