Chemistry, 13.07.2019 18:30 sindy35111
Given that the freezing point depression constant for water is 1.86°c kg/mol, calculate the change in freezing point for a 0.907 m sugar solution.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1


You know the right answer?
Given that the freezing point depression constant for water is 1.86°c kg/mol, calculate the change i...
Questions







Mathematics, 16.10.2020 05:01




History, 16.10.2020 05:01


History, 16.10.2020 05:01

Engineering, 16.10.2020 05:01


Mathematics, 16.10.2020 05:01



Mathematics, 16.10.2020 05:01

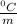
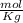 )
)