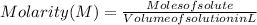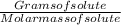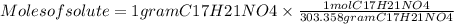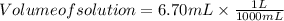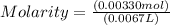Chemistry, 13.07.2019 20:00 myrkaxsanchezz
The free-base form of cocaine has a solubility of 1.00 g in 6.70 ml ethanol (ch3ch2oh). calculate the molarity of a saturated solution of the free-base form of cocaine in ethanol.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:50
2points why do scientists need governmental funding? o a. government politicians ask all the important scientific questions. o b. scientists have to pay taxes to the government on the money they make. o c. the cost of doing scientific research can be very high. o d. the government is controlled by scientists. submit
Answers: 3

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1


Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2
You know the right answer?
The free-base form of cocaine has a solubility of 1.00 g in 6.70 ml ethanol (ch3ch2oh). calculate th...
Questions



Biology, 21.04.2020 22:03

Social Studies, 21.04.2020 22:03

Physics, 21.04.2020 22:03


Mathematics, 21.04.2020 22:03

Mathematics, 21.04.2020 22:03


Mathematics, 21.04.2020 22:03




Mathematics, 21.04.2020 22:03



Mathematics, 21.04.2020 22:03

Chemistry, 21.04.2020 22:03

Mathematics, 21.04.2020 22:03