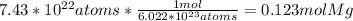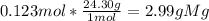Chemistry, 13.07.2019 20:30 payshencec21
How many grams of magnesium (mg, 24.30 g/mol) are in 7.43x10^22 atoms of mg?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1


Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
How many grams of magnesium (mg, 24.30 g/mol) are in 7.43x10^22 atoms of mg?...
Questions

Computers and Technology, 24.06.2020 15:01

Mathematics, 24.06.2020 15:01

Mathematics, 24.06.2020 15:01







Mathematics, 24.06.2020 15:01

Mathematics, 24.06.2020 15:01


Mathematics, 24.06.2020 15:01

Mathematics, 24.06.2020 15:01

Chemistry, 24.06.2020 15:01

History, 24.06.2020 15:01

Mathematics, 24.06.2020 15:01


Mathematics, 24.06.2020 15:01

Biology, 24.06.2020 15:01

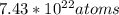
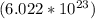 of formula units.
of formula units.