Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2


Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1
You know the right answer?
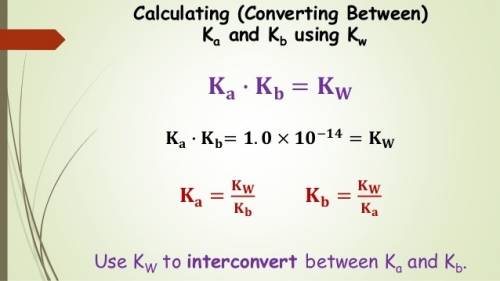
The acid dissociation constant ka for an unknown acid ha is 4.57 x 10^-3 what is the base dissociati...
Questions

History, 12.12.2020 16:50



Mathematics, 12.12.2020 16:50

Advanced Placement (AP), 12.12.2020 16:50


Mathematics, 12.12.2020 16:50

Physics, 12.12.2020 16:50


Mathematics, 12.12.2020 16:50


English, 12.12.2020 16:50

Social Studies, 12.12.2020 16:50


Mathematics, 12.12.2020 16:50


Mathematics, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50


English, 12.12.2020 16:50