
Chemistry, 15.07.2019 09:00 Jenniferwolf
How many moles of oxygen react with hydrogen to produce 22.8 mol of h2o? the balanced equation is as follows: 2h2 + o2 → 2h2o how many grams of co2 are produced if 35.48 mol of hcl are reacted according to this balanced chemical equation? caco3 + 2hcl → cacl2 + co2 + h2o

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle i need : ( asap i go it never mind
Answers: 2

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 07:30
Aradio signal from a gps satellite take only about 0.067 seconds to reach a gps reciever. if the speed of light is about 300,000km/s, then approximately how far away is the reciever from from the satellite?
Answers: 1

Chemistry, 22.06.2019 08:00
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1
You know the right answer?
How many moles of oxygen react with hydrogen to produce 22.8 mol of h2o? the balanced equation is a...
Questions

Social Studies, 13.11.2020 01:00

Chemistry, 13.11.2020 01:00


Mathematics, 13.11.2020 01:00

Biology, 13.11.2020 01:00




English, 13.11.2020 01:00





Mathematics, 13.11.2020 01:00


History, 13.11.2020 01:00



Mathematics, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00

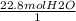 *
* 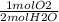 =
=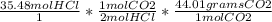 =
=

