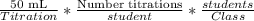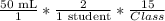Ateacher wants to figure out the amount of a solution that is needed for a titration experiment in a class lab. the class has 15 students, and each experiment needs 50 ml of solution. if each student will perform the experiment two times, how many ml of solution will the teacher need for the whole class? 375 ml 750 ml 1500 ml 3000 ml

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 13:50
Amap that uses a range of colors and shading to represent the elevation, depth, or landscape of specific features on earth is a/an map.
Answers: 3

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1

You know the right answer?
Ateacher wants to figure out the amount of a solution that is needed for a titration experiment in a...
Questions

English, 17.12.2020 04:50

Mathematics, 17.12.2020 04:50


Mathematics, 17.12.2020 04:50

Mathematics, 17.12.2020 04:50



Mathematics, 17.12.2020 04:50

Mathematics, 17.12.2020 04:50

English, 17.12.2020 04:50

Mathematics, 17.12.2020 04:50


Engineering, 17.12.2020 04:50



Mathematics, 17.12.2020 04:50

Mathematics, 17.12.2020 04:50

Advanced Placement (AP), 17.12.2020 04:50

History, 17.12.2020 04:50

Mathematics, 17.12.2020 04:50